Elidel and Protopic will contain a “black box” warning about a possible risk of cancer, the U.S. Food and Drug Administration announced Jan. 19, 2006.
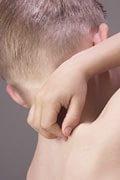
The FDA said it had received 78 reports of cancer – including lymphoma and skin cancers – among patients who used the creams to treat eczema, a skin disorder that causes a rash-like inflammation and itchy, red skin.
Eczema is one of the most common skin disorders found among infants and children, affecting 10-15 percent of the childhood population. An estimated 10 million people have used Elidel cream ( pimecrolimus), made by Novartis , and Protopic ointment, made by Astellas Pharma, Inc., formerly Fujisawa Healthcare.
If you or someone you know suffered a serious cancer after using Elidel or Protopic, you may want to contact an Elidel attorney or Protopic attorney for a free evaluation of your case.
A “black box” warning is the most serious type of warning for prescription drug labeling. While no studies have conclusively linked Elidel or Protopic to skin cancer, the FDA noted the 78 reports of cancer received by October 2005 plus the results of earlier animal studies suggesting a correlation were enough to issue the warning to doctors and patients.
“We are taking steps to ensure that healthcare providers and patients are aware of the possible long-term risks of these products so they will be used appropriately,” said Dr. Steven Galson, director of the FDA’s Center for Drug Evaluation and Research.
In February 2005, an FDA advisory committee recommended label warnings for Elidel cream and Protopic ointment after animal tests showed an increase of cancer associated with the drugs. The FDA said the manufacturers are currently conducting studies on the drugs but it could take “many years” to complete the research.
Elidel cream and Protopic ointment are intended to be used for short periods, the FDA noted. It advised patients to call their doctors if symptoms do not improve within six weeks of treatment or if the condition worsens or infections develop.
The new drug labeling will also note that Elidel and Protopic are recommended as second-line treatment, meaning that they should be used only after other topical medicines are tried first. The labels also will warn that Elidel and Protopic are not recommended for children younger than two years old.
Additionally, pharmacists will be required to provide patients with a Medication Guide on how to use the drugs safely.
Kline & Specter, PC, with some 30 attorneys (several of whom are also highly regarded doctors), has broad expertise and experience in pursuing pharmaceutical injury claims. Our law firm, based in Philadelphia and serving the Pennsylvania-New Jersey-Delaware region as well as clients nationwide, has produced superb results in a large number of personal injury cases. Contact an Elidel lawyer or Protopic lawyer today.
News
- FDA, drug makers knew of cancer risk before Elidel and Protopic skin creams hit the market, finds media investigation.
- Eczema drugs in USA Today


