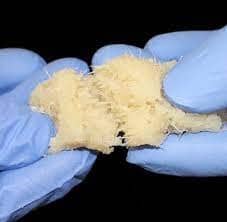
FiberCel, a product used to help mend bones, may have caused tuberculosis in as many as 113 patients across 20 states. A TB-contaminated lot was distributed this past spring and has been recalled by maker Aziyo Biologics.
If you or a loved one contracted tuberculosis after being treated for spinal surgery or a bone fracture, you may have been treated with FiberCel and could have grounds for a lawsuit.
Kline & Specter, PC, is representing patients who contracted tuberculosis through medical procedures and is accepting additional clients at this time. With more than 50 attorneys, five of whom are also medical doctors, our law firm has the experience to handle such complex medical product liability cases. The firm has successfully represented clients in many medical device and drug product cases.
Kline & Specter lawyers have won billions in product liability and personal injury lawsuits. Our doctor-lawyers give us the capability to quickly and expertly assess new cases. We offer free case evaluations.
Defendants in one such case, in Delaware, are Aziyo Biologics and Medtronic, which shipped the contaminated product, a regenerative bone “putty” that is made from human cadaver cells. The contaminated FiberCel was used in 113 patients in 37 facilities across 20 states, with the most reported in Delaware and Indiana.
Health officials said patients who received FiberCel from the bad batch should be treated even if they do not show symptoms of tuberculosis, which include a bad and long-lasting cough, chest pain, coughing up blood, weakness, fatigue, weight loss, fever and chills. TB can be fatal if not treated.
Tuberculosis is relatively rare, with only about 9,000 cases reported annually in the United States.

